AMOLF and Utrecht University have been awarded a 1.9 M€ research project by the Dutch Research Council (NWO) Key Technologies 2020-2023 programme to develop radically new catalytic reactors using plasmonic systems. The project entitled “Sensing and Steering Catalytic Reactions with Light” (CatLight) is a multidisciplinary collaboration between AMOLF (Alarcon-Llado, Albrecht, Ehrler, Garnett and Polman), Utrecht University (Weckhuysen, Hutter, Rabouw), and industrial partners BASF, ExxonMobil, Shell, Toyota Motor Europe, DENSsolutions, and Delmic.
The focus of the CATLight research project is to gain fundamental understanding and to develop new technologies that deliver targeted heat in industrially relevant catalyst systems with unprecedented spatio-temporal accuracy. This will create a promising new platform for efficient catalysis by using light and nanophotonic concepts. The new optically-driven catalysis technology will thus contribute to create more sustainable ways to efficiently drive important chemical reactions. The smarter use of energy and active thermal management enabled by photonic architectures developed in this research project will represent a paradigm shift for modern catalysis towards sustainable chemistry research.
Research plans
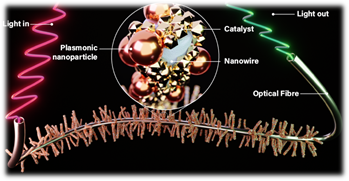
The consortium will demonstrate an entirely new way to make commercially relevant catalytic processes more active, selective and energy efficient. It will do so by combining the mature control over light-matter interactions at AMOLF, the unique in-situ and operando spectroscopy for investigating catalytic processes at Utrecht University and the know-how of the chemical and analytical industry partners.
CatLight introduces the use of intense light pulses to steer chemical reactions at the catalytically active sites that are locally heated by absorption of optical radiation at plasmonic nanoparticles. A 3D network of optical waveguides decorated with plasmonic nanoreactors guides intense light pulses to the catalytically active sites and is embedded in a reaction vessel. Simultaneously, optical excitation provides key thermal and spectroscopic fingerprints of the reactive species involved in the chemical pathways as they proceed in space and time.
“The use of light offers opportunities to control chemical reactions in ways that can never be obtained with conventional heating. For instance, light-induced heating with pulsed illumination can create fast temporal energy landscapes that cannot be achieved with conventional heating, enabling chemical reaction pathways that are currently impossible.”
Dr. Esther Alarcon Llado (consortium leader), AMOLF
“The large operational regime offered here by plasmonic-heating can provide a new breakthrough in the field of catalysis. This, combined with time-resolved spectroscopy will open the possibility to learn about elementary reaction limiting steps and inspire novel (or more efficient) catalyst designs. The impact of local thermal spikes on catalyst-gas interface dynamics is not well-known. By developing experimental evidence on plasmon-based ultrafast heating in nano-reactors, we will explore a new branch in light-based catalysis.”
Prof. Bert Weckhuysen, Utrecht University

